Sudan virus disease (SVD)
Ongoing outbreak in Uganda
On 20 September 2022, Uganda declared an outbreak of Sudan virus disease (SVD) caused by Sudan virus (SUDV) after samples tested positive for Sudan virus (SUDV) following six suspicious deaths in the country’s central Mubende District.
So far, the outbreak has affected Bunyangabu Kagadi, Kampala, Kasanda, Kyegegwa, Masaka, Mubende, Jinja and Wakiso districts. The virus has spread to capital Kampala. It is the first time since 2012 that Uganda has experienced an SVD outbreak.
Preliminary genomic sequencing by the Uganda Virus Research Institute indicates that the current outbreak is closely related to Nakisamata Sudan ebolavirus strain that emerged in Luwero District, Uganda in May 2011. Investigations are underway to determine the index case and the exact origins of the ongoing outbreak.
There have been seven previous outbreaks of the Sudan ebolavirus, with four occurring in Uganda and three in Sudan. In 2019, Uganda country experienced an outbreak of Ebola virus disease (EVD) caused by Zaire ebolavirus following imported cases from neighbouring Democratic Republic of the Congo which was battling a large epidemic in its north-eastern region.
Case fatality rates of the Sudan virus have varied from 41% to 100% in past outbreaks.
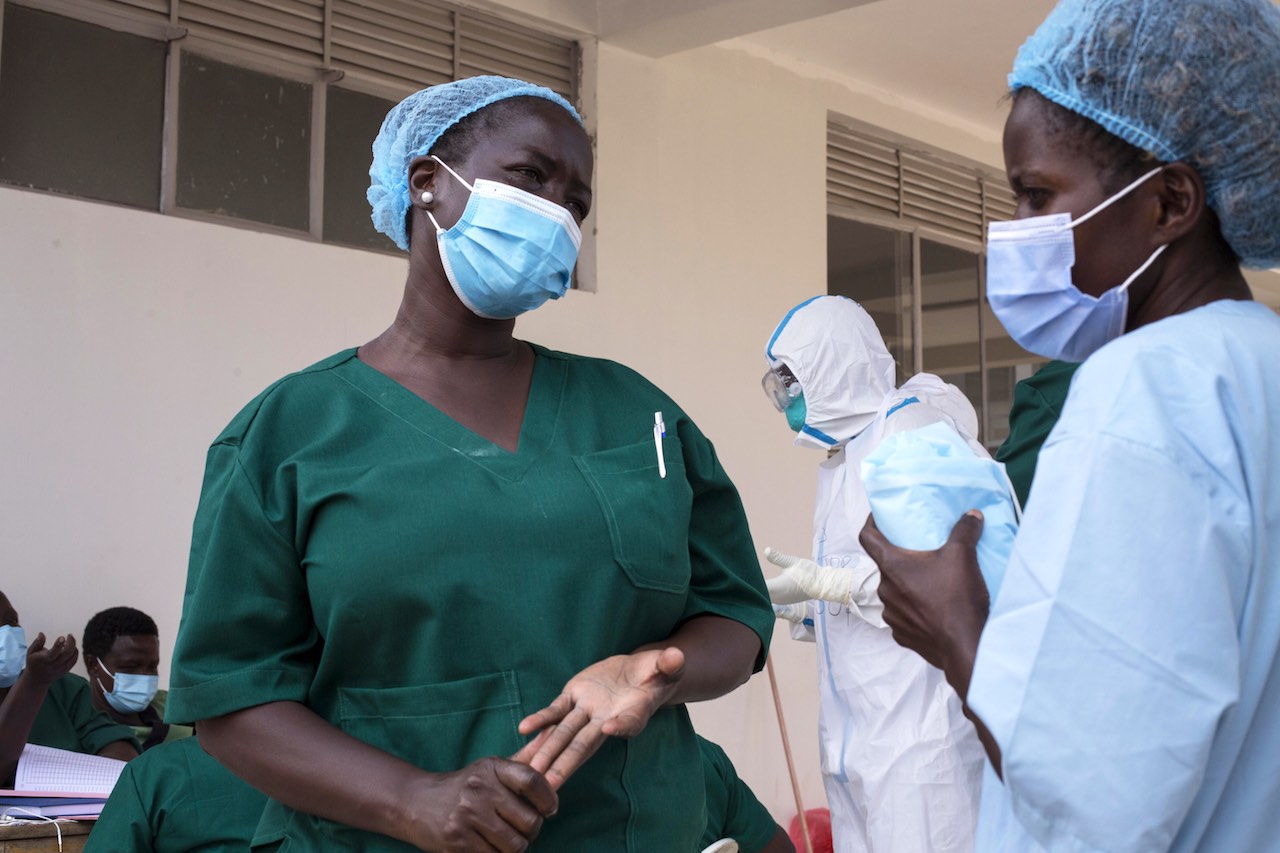
Response
As no effective vaccine against the Sudan ebolavirus has been licenced yet, Ugandan health authorities have focused on supportive care for confirmed cases alongside stepping up testing, surveillance, infection prevention and control, as well as collaborating with communities to support disease prevention measures.
WHO and partners are supporting the government bolster all the key aspects of Ebola control. The Organization is delivering medical supplies and providing logistics and had deployed experts to work with the national response teams.
Key measures being reinforced include testing, infection prevention and control including supplies such as personal preventive equipment to health workers and other frontline responders. Public awareness campaigns as well as safe and dignified burials for any deaths linked to Ebola are also being stepped up. Treatment centres have been set up with additional beds, contact tracing, including by working with village health teams, has been enhanced and monitoring of cases significantly improved since the onset of the outbreak.
To support the response and help stem potential cross-border spread of the virus, health authorities from neighbouring countries have agreed on joint countermeasures that include bolstering disease surveillance, contact tracing and monitoring, prompt alert notification, information sharing and joint trainings of emergency responders, as well as carrying out simulation exercises to enhance preparedness and response.
Vaccination
There are now three candidate vaccines (one from Sabin Institute, University of Oxford & SII and the Merck vaccine donated to IAVI) that have been considered by WHO and the MOH Uganda for trial against Sudan ebolavirus, which are in different stages of development.
Ring vaccination to evaluate the efficacy and safety of these vaccines will be conducted as soon as the country receives the vaccine doses.
Overall preparedness to start vaccination is at 82%; ultracold chain already secured and handed over to WCO. Principal Investigator and sub-investigators selected and engaged by WHO. Good clinical practices and ring vaccination trainings completed. Space for Kampala and Mubende trials identified and secured, renovations ongoing. Local IRB feedback on protocol received and comments addressed.