Fact sheet
- Seasonal influenza is an acute viral infection that spreads easily from person to person.
- Seasonal influenza viruses circulate worldwide and can affect people in any age group.
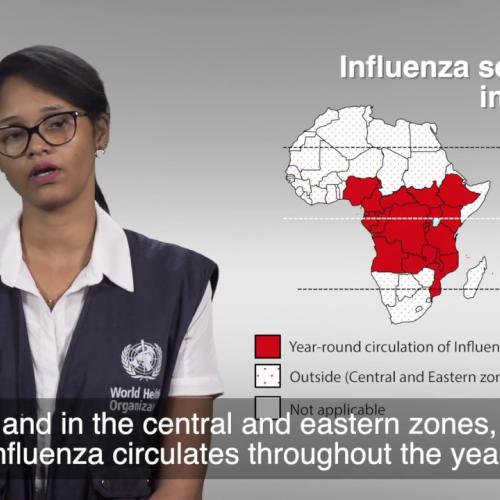
- In temperate climates, seasonal epidemics occur mainly during winter while in tropical regions, influenza seasonality is less obvious and epidemics can occur throughout the year.
- Seasonal influenza is a serious public health problem that causes severe illness and death in high risk populations.
- An influenza epidemic can take an economic toll through lost workforce productivity and strained health services.
- Influenza vaccination is the most effective way to prevent disease.
- Antiviral drugs are available for treatment, though influenza viruses can develop resistance to the drugs.
Seasonal influenza is characterized by a sudden onset of fever, cough (usually dry), headache, muscle and joint pain, severe malaise (feeling unwell), sore throat and a runny nose. The cough can be severe and can last 2 or more weeks. Most people recover from fever and other symptoms within a week without requiring medical attention. But influenza can cause severe illness or death especially in people at high risk (see below). The time from infection to illness, known as the incubation period, is about 2 days.
Yearly influenza epidemics can seriously affect all populations, but the highest risk of complications occur among pregnant women, children aged 6–59 months, the elderly, individuals with specific chronic medical conditions such as HIV/AIDS, asthma, and chronic heart or lung diseases, and health-care workers.
Seasonal influenza spreads easily, with rapid transmission in crowded areas including schools and nursing homes. When an infected person coughs or sneezes, droplets containing viruses (infectious droplets) are dispersed into the air and are spread to persons in close proximity who breathe these droplets in. The virus can also be spread by hands contaminated with influenza viruses. To prevent transmission, people should cover their mouth and nose with a tissue when coughing, and wash their hands regularly.
In temperate climates, seasonal epidemics occur mainly during winter, while in tropical regions, influenza may occur throughout the year, causing outbreaks more irregularly.
Illnesses range from mild to severe and even death. Hospitalization and death occur mainly among high-risk groups. Worldwide, these annual epidemics are estimated to result in about 3 to 5 million cases of severe illness, and about 250 000 to 500 000 deaths.
In industrialized countries most deaths associated with influenza occur among people age 65 or older. Epidemics can result in high levels of worker/school absenteeism and productivity losses. Clinics and hospitals can be overwhelmed during peak illness periods.
The effects of seasonal influenza epidemics in developing countries are not fully known, but research estimates indicate that 99% of deaths in children under 5 years of age with influenza related lower respiratory tract infections are found in developing countries.
The most effective way to prevent the disease is vaccination. Safe and effective vaccines are available and have been used for more than 60 years. Among healthy adults, influenza vaccine provides protection, even when circulating viruses may not exactly match the vaccine viruses.
However, among the elderly, influenza vaccination may be less effective in preventing illness but reduces severity of disease and incidence of complications and deaths. Vaccination is especially important for people at higher risk of serious influenza complications, and for people who live with, or care for, high risk individuals.
WHO recommends annual vaccination for:
- pregnant women at any stage of pregnancy
- children aged between 6 months to 5 years
- elderly individuals (aged more than 65 years)
- individuals with chronic medical conditions
- health-care workers.
Influenza vaccine is most effective when circulating viruses are well-matched with viruses contained in vaccines. Due to the constant evolving nature of influenza viruses, the WHO Global Influenza Surveillance and Response System (GISRS) – a network of National Influenza Centres and WHO Collaborating Centres around the world – continuously monitors the influenza viruses circulating in humans and updates the composition of influenza vaccines twice a year.
For many years, WHO has updated its recommendation on the composition of the vaccine that targets the 3 (trivalent) most representative virus types in circulation (two subtypes of influenza A viruses and one influenza B virus). Starting with the 2013–2014 northern hemisphere influenza season, a 4th component is recommended to support quadrivalent vaccine development. Quadrivalent vaccines include a 2nd influenza B virus in addition to the viruses in trivalent vaccines, and are expected to provide wider protection against influenza B virus infections. A number of inactivated influenza vaccines and recombinant influenza vaccines are available in injectable form. Live attenuated influenza vaccine is available as a nasal spray.
Antiviral drugs for influenza are available in some countries and may reduce severe complications and deaths. Ideally they need to be administered early (within 48 hours of onset of symptoms) in the disease. There are 2 classes of such medicines:
-
inhibitors of the influenza neuraminidase protein (oseltamivir and zanamivir; as well as peramivir and laninamivir which are licensed in several countries).
- M2 proton channel blockers adamantanes (amantadine and rimantadine), to which virus resistance has been frequently reported, limiting the effectiveness of treatment.
WHO monitors antiviral susceptibility among circulating influenza viruses to provide timely guidance for antiviral use in clinical management and potential chemoprophylaxis. Currently, the majority of circulating influenza viruses are resistant to the adamantanes and WHO recommends neuraminidase inhibitors as the first-line treatment for people requiring antiviral therapy.
WHO, through the GISRS network, in collaboration with other partners, monitors influenza activity globally, recommends seasonal influenza vaccine compositions twice a year for the Northern and Southern hemisphere influenza seasons, and supports Member States’ efforts to develop prevention and control strategies.
WHO works to strengthen national, regional and global influenza response capacities including diagnostics, antiviral susceptibility monitoring, disease surveillance and outbreak responses, and to increase vaccine coverage among high-risk groups and prepare for the next influenza pandemic.